Get the latest breaking news and trends about paid media with our newsletter. Stay updated on what is new in the world of PPC ads, Google Ads, Bing Ads and more and outrank your competition.
Get daily dose of marketing news in quick TL;DR style.
I love tl;dr Marketing because I can get all the latest SEO news and trends in one spot without having to read lengthy articles. I really look forward to the daily emails to see what's new in our industry!
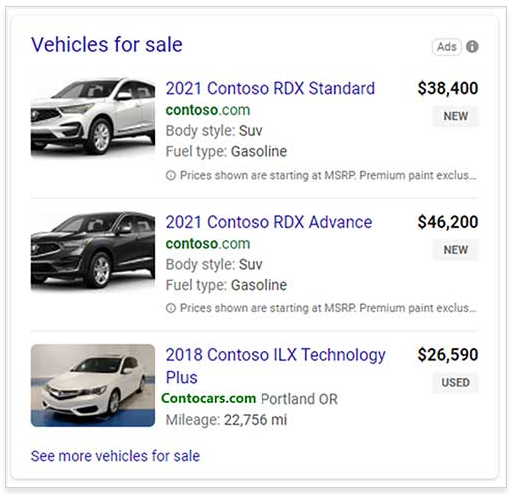
Microsoft Advertising Automotive Ads Rolling Out To All Advertisers Globally
about.ads.microsoft.comMicrosoft Advertising is making Automotive Ads generally available to all advertisers globally in the coming weeks!
You can now use Dynamic Remarketing for Travel, Automotive, and Events.
In-market Audiences is now available in more markets
Latin America: Aruba, Bahamas, Bolivia, Cayman Islands, Costa Rica, Dominica, Dominican Republic, Ecuador, El Salvador, French Guiana, Guatemala, Guyana, Haiti, Honduras, Martinique, Montserrat, Panama, Paraguay, Puerto Rico, Trinidad and Tobago, and Uruguay.
Asia Pacific: Bangladesh, Brunei, Fiji, French Polynesia, Guam, Maldives, Mongolia, Nepal, New Caledonia, Papua New Guinea, and Sri Lanka.
FLEDGE is a Privacy Sandbox proposal for remarketing and audiences. It’s designed so that it can’t be used by third parties to track user browsing behavior across websites. Google will begin testing the API on AsSense accounts on August 28.
You can learn more about the API here.


 Web Stories
Web Stories